Neuroscientist Helen Murray’s special expertise with a tiny part of the brain is helping unravel the mysteries of Covid-19’s neurological effects.
For weeks, University of Auckland neuroscientist Helen Murray pored over scans of brain tissue from people who had died from Covid-19.
She was collaborating with scientists in a government research agency in the United States, the National Institutes of Health (NIH).
The focus of the research: what exactly does Covid-19 do to the brain?
While Covid-19 is primarily a respiratory disease, it’s been estimated that a third of hospitalised patients have neurological symptoms. The most common are dizziness and headache but stroke may occur in as many as 5% of hospitalised cases, initial studies suggest.
“The most common severe neurological effects, stroke and encephalitis, can have life changing implications, especially for someone who is young and was previously healthy,” says Dr Murray, who’s a research fellow at the Centre for Brain Research in Grafton.
In the U.S., researchers last year obtained the brain tissue of 19 deceased Covid-19 patients from the pathology department of the University of Iowa College of Medicine and the Office of the Chief Medical Examiner in New York City. The patients, aged from 5 to 73, died between a few hours and two months after reporting Covid-19 symptoms. Their risk factors had included diabetes, obesity and cardiovascular disease. Three collapsed suddenly.
The scientists were grateful for the brain samples, which were difficult to obtain in the early days of the pandemic because of the dangers of infection during autopsies.
Dr Murray’s involvement was because of her expertise in dealing with the olfactory bulb, the tiny part of the brain that enables us to smell. It’s the only part of the brain connected, via the nose, to the outside world, making it vulnerable to pollutants and viruses.
The neuroscientist had existing ties with the NIH from a joint project investigating the role of the olfactory bulb in Alzheimer’s disease, another disease involving loss of smell. In that project, Dr Murray worked with samples from the Neurological Foundation Human Brain Bank at the Centre for Brain Research, developing skills in handling and examining brain tissue that are especially niche, since many of the world’s brain banks do not routinely collect olfactory bulbs.
In the U.S., the NIH scientists, who are based near Washington DC, used especially sensitive magnetic resonance imaging (MRI) machines to scan tissue from the deceased patients’ brain stems and olfactory bulbs, two places known for Covid-19 effects. They saw evidence of bleeding in these two brain regions.
Travel restrictions prevented Dr Murray from going to the U.S. as she had in the past, so she helped via Zoom and email, advising on preparations and then examining the results.
In the U.S., the scientists examined the regions of bleeding in more detail by adding fluorescent antibodies to the brain tissue and looking through microscopes. The fluorescent antibodies bound to proteins to show where blood vessels were thinner than normal and blood proteins had leaked into brain tissue. Immune cells surrounded the leaks, indicating an inflammatory response.
This type of damage is typically associated with stroke and neuroinflammatory diseases such as Alzheimer’s disease, Parkinson’s disease, and multiple sclerosis.
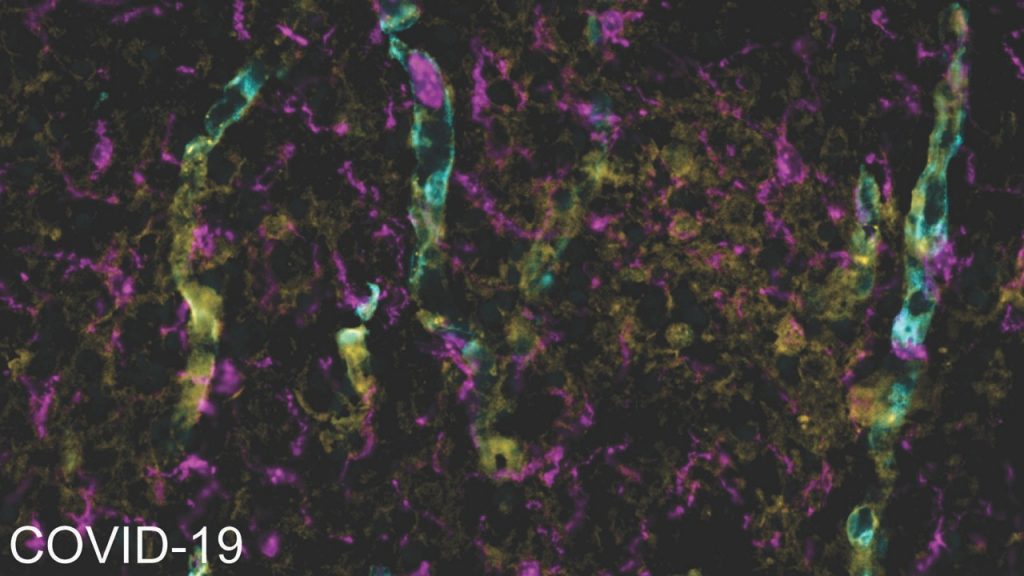
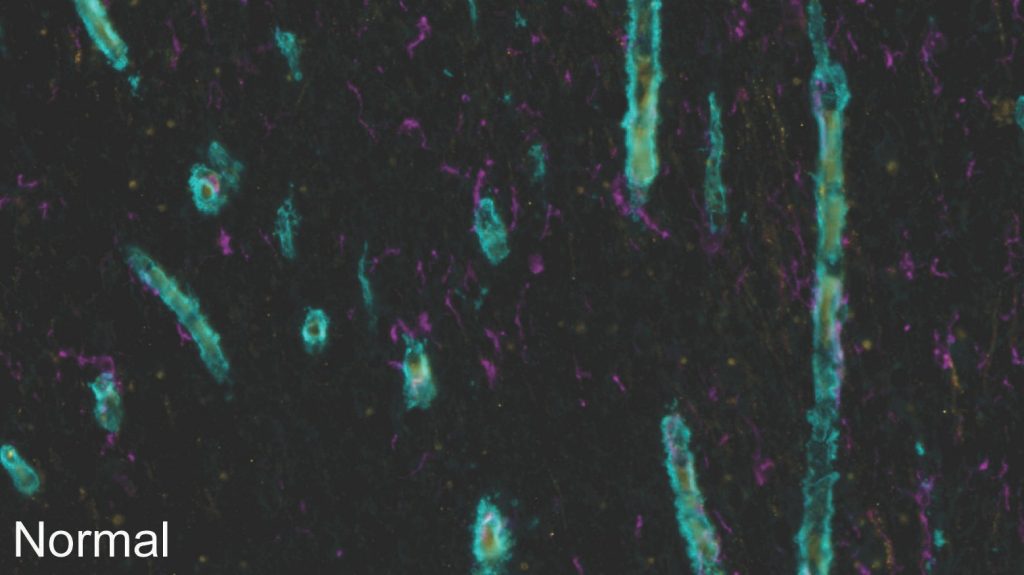

 In the images above, blood vessels are greeny-blue, microglia (immune cells) are magenta and the blood protein fibrinogen is yellow. In the brain tissue of a Covid-19 patient, the fibrinogen has leaked out of the blood vessels and the extra microglia indicate an immune response.
In the images above, blood vessels are greeny-blue, microglia (immune cells) are magenta and the blood protein fibrinogen is yellow. In the brain tissue of a Covid-19 patient, the fibrinogen has leaked out of the blood vessels and the extra microglia indicate an immune response.
Back in New Zealand, working from home in Botany because of lockdown, Dr Murray wrestled with giant files in her inbox, which were the brain scans in high-resolution, microscopic detail. What she saw surprised her.
“It was striking. I’ve looked at tissue from Alzheimer’s patients for years now, but I have never seen changes as severe as these in the olfactory bulb.”
Scientists had hypothesized that a loss of smell could be caused by SARS-CoV-2, the virus that causes Covid-19, getting into the olfactory bulb. However, Dr Murray and her colleagues saw no sign of a direct attack. Instead, the changes in the brain looked as if they could be part of a broader inflammatory response.
Could the changes contribute to a loss of smell? “It’s possible,” says Dr Murray. “I would be very surprised if there wasn’t an effect, but we still don’t know.”
A recent study out of Harvard Medical School focused on a different factor that could sabotage smell: the infection by the virus of cells in the nose that support odour-detecting neurons.
There’s so much more to learn – including following up on the speculation that Covid-19 patients could have an increased risk of later developing Parkinson’s, Alzheimer’s or multiple sclerosis or suffering stroke. It’s noteworthy that the Spanish flu pandemic of 1918 was followed by a surge in cases of Parkinson’s. The world’s total of 115 million Covid-19 cases as of March highlights the potential scale of any long-term knock-on effects.
In the U.S., NIH has launched a database to collect information from clinicians about Covid-19-related neurological conditions as well as Covid-19 effects on pre-existing neurological conditions.
Dr Murray and her collaborators – led by Dr Avindra Nath, of the NIH – plan to keep investigating. “The next study will focus on the specific olfactory bulb structures involved in odour detection,” says Murray. “We want to understand if the inflammation and the leaks from blood vessels are near these structures and contribute to the loss of smell.”
Dr Murray is a Health Education Trust post-doctoral research fellow at Brain Research New Zealand, based at the University of Auckland and holds a non-clinical collaborator appointment at the National Institute of Neurological Disorders and Stroke (NINDS) at the National Institutes of Health, USA.
This article was originally published by the University of Auckland.



